Does Nickel Rust? – Concept, Factors, Misconceptions, and More
Table of Contents
ToggleUnderstanding Nickel
Nickel is a transitional metal whose symbol is Ni and atomic number 28. It is shiny, silvery white and is non-corrosive. You will observe that it is malleable, ductile, and medium hard. It reacts slowly with water and Oxygen to create protective oxide layer. Nickel is found in coins, electronics, batteries, and plumbing fixtures. Nickel alloys are materials that make things stronger and are essential in the modern industry.
Rust vs Corrosion: The Science Behind Nickel
The rusting process occurs when iron combines with oxygen and moisture to form iron oxide, which can be Fe₂O3 or Fe3O4. Corrosion refers to any loss of the metal through a chemical reaction of the material with the surroundings. You can only see rust in iron and alloys. This often raises the question: does nickel rust? Pure nickel does not rust since it does not have iron. Rather, nickel produces nickel oxide, nickel hydroxide or nickel carbonate. These compounds are able to form a protective passive layer, which retards further attack and enhances durability in moderate conditions. Passive protection is contrasted to destructive corrosion, which does not allow the constant loss of the material.

Feature | Nickel Rusting | Nickel Corrosion | Iron Rust |
Definition | Impossible | Possible | Possible |
Primary Compound | – | NiO, Ni(OH)₂, NiCO₃ | Fe₂O₃ |
Appearance | – | Tarnish, pits | Red-brown flaky |
Protective Layer | – | Yes (oxide) | No |
Occurs in Water | No | Rare (pH, Cl⁻) | Yes |
Occurs in Salt Water | No | Yes (pitting) | Yes |
Rate of Degradation | 0 | Low to moderate | High |
Does Nickel Rust?
Nickel does not rust due to the fact that rust is formed on iron, so the question of “will nickel rust” does not apply in the traditional sense. It has the ability to corrode in extreme conditions. Nickel defends itself by coating itself in a passive oxide layer of around 2 5 nm. This is a non-porous film that prevents oxygen and moisture. Its excellent corrosion resistance is beneficial to you in numerous applications. Nickel has much better resistance to corrosion. It also does better than galvanized steel, zinc, and magnesium. Nickel remains unchanged in maritime, chemical plants and moist air.

Factors Making Nickel Rust Resistant
- Alloying: Nickel alloys are reinforced with chromium, molybdenum, and copper. You enhance resistance to corrosion in challenging conditions. The microstructure is refined by these elements and decreases surface reactivity. That assist you in increasing service life.
- Surface Finish: Nickel has smooth surface so there is no doubt to say does nickel rust, as nickel has a smooth surface that restricts the points of corrosion initiation. It decrease dampness and dirt accumulation. Smooth finishes are easy and effective to clean. This insurance helps in long-term performance.
- Passivation: Nickel develops a protective oxide layer in an oxygen rich environment. This passivation protects the metal against additional attack. It is helpful when the surface is not covered. The barrier regenerates itself.
- Low Reactivity: Nickel is a low reactive element in comparison to most metals. You undergo slower oxidation under normal circumstances. The stability assists components to withstand heat and moisture. In strenuous industrial and marine environments.
- Chemical Stability: Nickel is chemically stable in most acids and bases. It is reliable in terms of performance. Its inertness decreases aggressive response and surface damage. In various real-world uses.
Oxidized Nickel vs Rusted Nickel
Nickel develops a thin layer of oxide which helps in providing protection against any further corrosion. This is predominantly nickel oxide layer that encloses the metal and restricts the contact of oxygen. Conversely, iron rusts, and it is porous and flaky. The rust continues to spread and undermines the structure in the long run. NiO (nickel oxide) is gray or black, and Fe 2O 3 (rust) is red-brown. Due to this stable barrier, you increase the life of nickel in the outdoor and marine environment. Such natural protection saves a lot of maintenance in the long run.

Nickel Corrosion: Conditions and Mechanisms
- Chloride Ions: Chloride ions are highly aggressive against nickel surfaces particularly in seawater and salty environments which develop pitting corrosion. Localized damage can be minimized by exposure reduction and protective coating.
- Strong Acids and Reducing Agents: People also wonder, does nickel rust in the presence of strong acids such as HCl and sulfuric acid, and reducing agents. Yes, these acids may dissolve the passive layer of this metal. Concentration and temperature should be regulated to ensure corrosive resistance in severe conditions.
- Contaminants in Nickel: Contaminants like iron, sulfur and carbon undermine the protective layer of nickel by forming microstructural flaws. You enhance performance by choosing high-purity grades and checking composition prior to critical applications in use.
- High Temperature Effects: Nickel passive layer becomes porous and thicker at high temperatures exceeding 600 C, thereby decreasing protection. You ought to observe thermal exposure and design systems which avoids excessive overheating and structural degradation.
Nickel Alloys and Their Corrosion Resistance
Alloy/Metal | Major Composition | Corrosion Resistance Advantage | Corrosion Rate in Seawater (mm/year) | Typical Applications |
Pure Nickel (99.99%) | Ni ≥ 99.99% | Excellent resistance in neutral & fresh water | 0.005–0.01 | Electronics, thermocouples, chemical vessels |
Monel 400 | 65% Ni, 34% Cu, 1% Fe | Superior seawater & reducing acid resistance | 0.002–0.005 | Marine valves, propeller shafts |
Inconel 625 | 59% Ni, 21.5% Cr, 9% Mo | Exceptional chloride, pitting, high-temp resistance | <0.001 | Offshore oil rigs, chemical reactors |
304 Stainless Steel | 18% Cr, 8% Ni, 74% Fe | Good general corrosion resistance | 0.01–0.02 | Kitchen appliances, architectural trim |
316 Stainless Steel | 16–18% Cr, 10–14% Ni, 2–3% Mo | Improved chloride resistance vs 304 | 0.005–0.01 | Marine equipment, chemical tanks |
Common Misconceptions About “Rust” on Nickel
Misconception 1: “Does nickel plated steel rusted.”
Although it is believed that nickel-plated steel is easy to rust, you must look at the underlying metal first. Nickel creates a shield that is impervious to moisture and oxygen. In cases of corrosion, it normally begins under broken coating, and the steel is exposed to the external environment.
Misconception 2: “Nickel turns brown.”
People often wonder does nickel rust, but when nickel may darken over time, this discoloration should not be mistaken with rust. Real rust involves the combination of iron and oxygen. On the surface, tarnish compounds develop, i.e. nickel sulfide or nickel carbonate, particularly in contaminated air, moisture, or exposure to chemicals.
Misconception 3: “Shower nickel rusted.”
Shower fixtures are frequently exposed to constant moisture and salts, thus spots or pits may be observed. Water chloride ions attack the surface and produce localized pitting corrosion. This corrosion ruptures nickel coatings without rusting, and ventilation decreases the danger.
To avoid corrosion, you need to focus on good maintenance and frequent cleaning to avoid damage to the protective surfaces. The use of durable finishes like lacquer provides an additional protective layer against moisture, oxygen and contaminants. Another thing is that you have to choose the right nickel alloys in your environment since the proper choice of material will increase long-term performance and durability considerably.
Nickel in Water and Outdoors
Freshwater
People also ask does nickel rust in freshwater? Nickel is resistant to corrosion in freshwater since it develops a stable nickel oxide. This protective coating helps to avoid rust and thus you may count on longevity in lakes, rivers and water systems.
Saltwater
Chloride ions in saltwater undermine the protective oxide layer. This injury may cause localized pitting of surfaces. You are supposed to check and guard materials that are near seawater to ensure that they are not deteriorated.
Outdoor Exposure
Normal environmental conditions do not tend to alter outdoor nickel. When the humidity is moderate and the pollutants are minimal, you perform. Excessive acids or continuous dampness may speed up the corrosion threat.
Corrosion rates and stability of materials are directly dependent on environmental conditions like temperature and the availability of oxygen and pH. Surface reactions may be quickened by higher temperatures, low oxygen levels, or extreme acidic and alkaline conditions, and risk deterioration.
Nickel Finishes: Rust and Tarnish Potential
- Satin Nickel: Satin Nickel does not rust under normal indoor and daily conditions. If you’re wondering does satin nickel rust? its durability is generally certain, but it can corrode over time when exposed to harsh moisture.
- Brushed Nickel: Brushed Nickel offers good corrosion protection to your fixtures and hardware. But you have to cover the exposed spots since scratches may expose steel and rust.
- Polished Nickel: Polished Nickel provides a bright finish and dependable rust resistance. It is important to clean it up regularly to maintain the shine and minimize the tarnish with time indoors.
- Black Nickel: Black Nickel has protective coating that prevents the formation of rust. The surface should be checked regularly as wear may decrease protection and raise tarnish.
Testing Nickel Corrosion Resistance
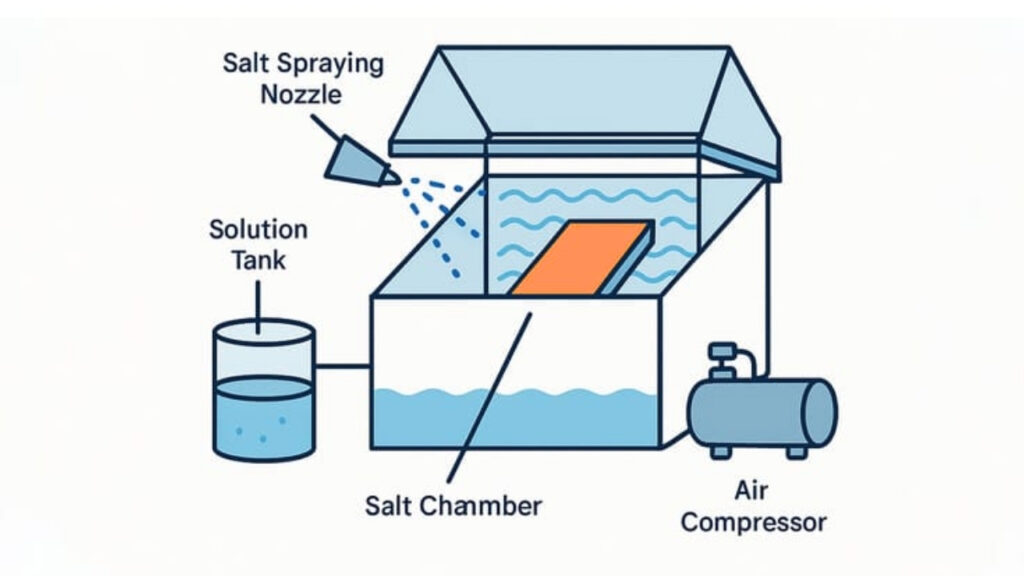
Salt Spray Test (ASTM B117)
A common question is: does nickel rust? This can be evaluated by exposing nickel plating to a 5% sodium chloride mist at 35°C. The test lasts 100 to 1,000 hours to test the duration. You see rusting and surface deterioration closely. You record findings to compare. This guarantees clarity in evaluation.
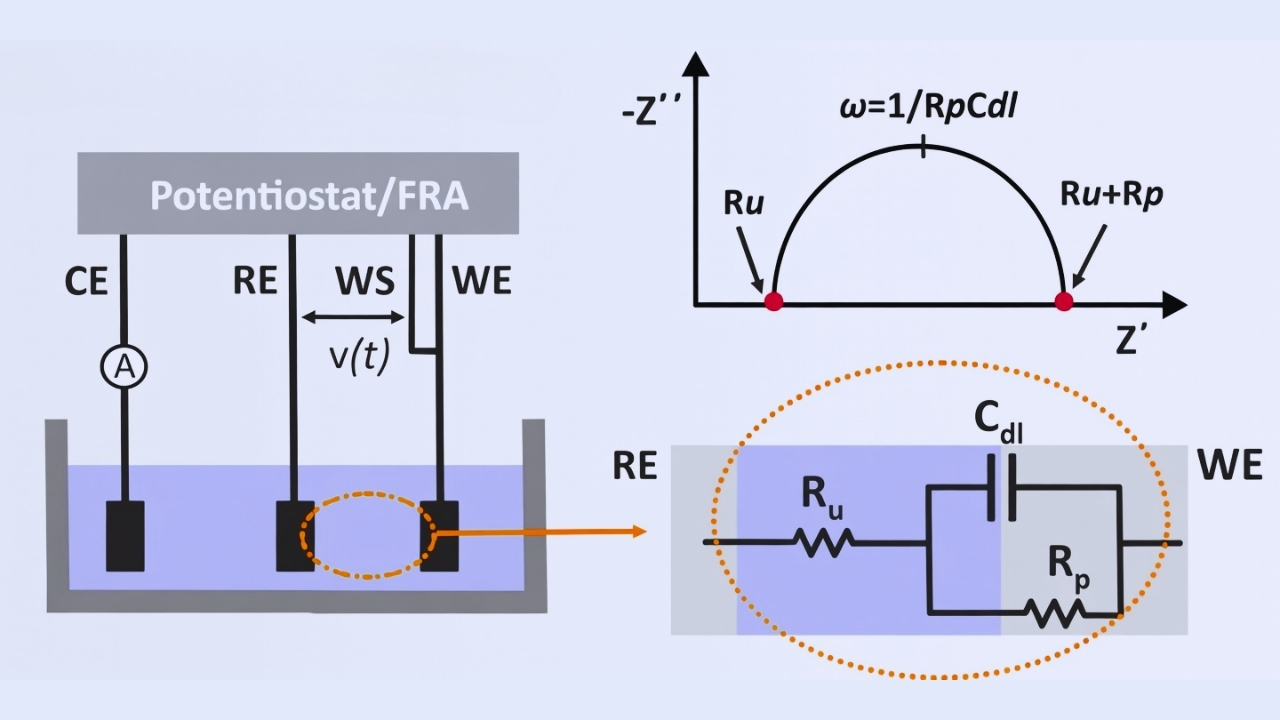
Electrochemical Impedance Spectroscopy (EIS)
You are analyzing the passive film of nickel surfaces using EIS. The method uses a small alternating signal and impedance response is measured. This aids you in determining coating integrity and corrosion processes. You measure the strength of the barriers without ruining the specimen.
Weight Loss Testing (ASTM G1)
You determine the rate of corrosion by weighing samples prior to and after exposure to certain environments. This technique measures the loss of materials with time. You compare performance and test durability. You choose appropriate nickel alloys in demanding use.

Industrial Applications Highlighting Nickel’s Non-Rusting Properties
- Marine Engineering: Marine engineering involves the use of nickel alloys in propellers, pumps, and offshore platforms. These materials enable you to fight seawater corrosion and lower long term maintenance expenses. Worldwide reliability.
- Chemical Processing: Nickel alloys that are used in chemical processing plants include Hastelloy and Monel. Strong acids can be safely handled with these materials. Confidently.
- Electronics: Batteries, terminals, and semiconductors made by electronics manufacturers contain ultra-pure nickel. Ultra pure performance assists you in attaining dependable conductivity and stable device running. In critical systems.
- Architecture: Nickel containing materials are used in architecture projects in facades and monuments across the world. These finishes allow you to reduce tarnish, reduce rust, and retain long term appearance —effectively addressing concerns like does nickel rust while maintaining durability and aesthetic appeal.
Best Practices for Maintaining Nickel Surfaces
Cleaning Techniques
Wipe off the dust and fingerprints of nickel items with warm water and mild soap. Wipe delicately with a soft cloth and dry properly to ensure no water spots and streaks.
Safe Natural Remedies
Use a paste of baking soda on stubborn stains and rub in circular motions. Lemon juice and salt will produce slight tarnish, but must be rinsed off to preserve surface.
Protective Coatings
The possibility of coating it with a clear lacquer to reduce air exposure and tarnish in the long term should be considered. In the case of industrial components, duplex nickel chrome systems should be specified to enhance corrosion resistance and life.
Regular Maintenance
Conduct frequent checkups to identify premature discoloration and wear on the surface. Wipe off small patches immediately to avoid corrosion and maintain the look in harsh conditions and routine working tasks. Be consistent in general.
Conclusion
Nickel is not rusted, as there is no iron in it, but it can decay under certain conditions. When considering whether does nickel rust, it is important to understand that rust specifically refers to iron oxidation, which nickel does not undergo. The natural oxide layer that it has offers great protection in water, air and most industrial environments. By alloying, surface finishing, and maintenance, you make great contributions to durability and performance. With the knowledge of the environmental factors, you can choose the correct nickel grade with a lot of certainty over the long term reliability and lower maintenance charges. Thus, nickel can be relied upon in demanding applications where conditions are well regulated in a variety of industry settings.
FAQs
Does Nickel Rust in Salt Water?
Nickel does not rust easily in most environments, although salt water exposes it to corrosion. Aged surface tarnish is to be anticipated with time, particularly without protective finishes or appropriate care in severe marine conditions.
Does Satin Nickel Rust?
Satin nickel is not immune to corrosion since it has a nickel finish coated on a different metal. You can safeguard it by washing delicately, dry surfaces, and not keeping surfaces wet in bathrooms or kitchen areas.
How Fast Will Nickel Rust?
Nickel does not rust rapidly as iron, but may oxidize slowly. The rate at which corrosion forms on the exposed surfaces is dependent on environmental factors like humidity, salt and acids.
Does Brushed Nickel Rust?
Brushed nickel has a textured surface, which conceals small scratches and fingerprints. Nevertheless, you should still take care of it, as the underlying substance is subject to corrosion in case the moisture is trapped too long.
Will Polished Nickel Rust?
Nickel is polished to give it a shiny look though it is susceptible to corrosion in harsh environments. You are supposed to wash it regularly and not to use harsh cleaners that destroy the surface layer.
Does Black Nickel Rust?
Black nickel is used to give decorative color by plating, although it may wear away. You minimize the risk of corrosion by ensuring that moisture is in contact with surfaces and that appropriate protective coating is used where feasible.
Does Cupro Nickel Rust?
Copper nickel or cupro nickel is a strong resistant corrosion alloy in the seawater. It is dependable in marine hardware, as the copper content enhances durability and reduces the rate at which rust develops.
Does Lead Nickel Rust?
Lead nickel alloys do not simply rust, but they do not readily do so depending on composition. Specific material data should be reviewed, as performance varies with conditions of exposure to the environment and manufacturing process.
Is Nickel More Corrosion Resistant Than Stainless Steel?
Nickel has good corrosion resistance, but typically stainless steel is more effective. You relate certain grades and settings to determine which material is effective in practice in terms of your project needs.
